開發多用途之兩步驟配體引導標功能化策略操控及探測細胞內原生蛋白
A versatile strategy to manipulate and probe native proteins in cellulo utilizing two-step ligand directing functionalization
計畫主持人:臺大-化學系 王宗興、中研院-生醫所 林宛蓁
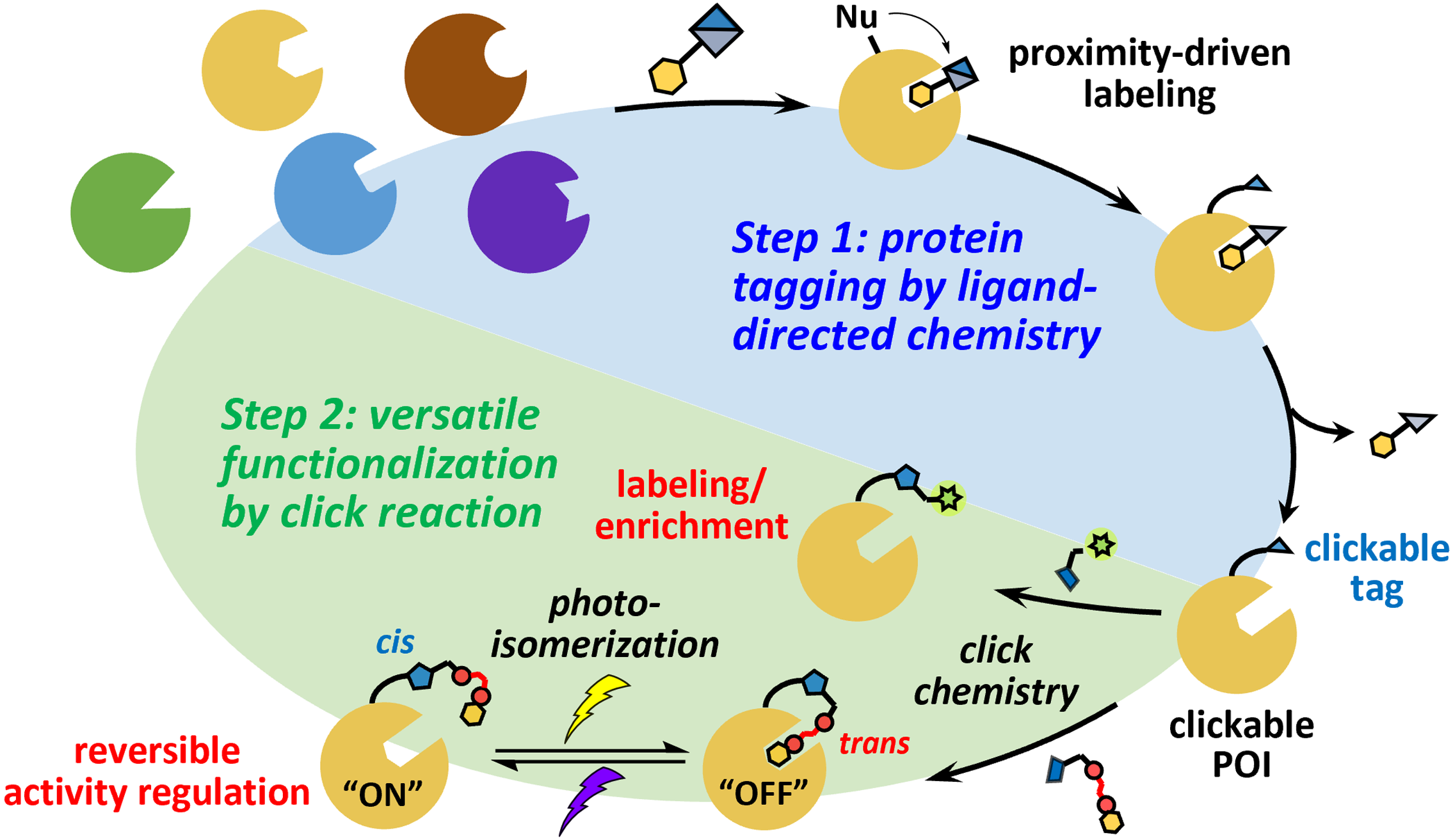
解析蛋白質在其天然環境中的生物運輸與功能相互作用,一直是生物學領域的重要挑戰。蛋白質的化學修飾有助於我們深入理解其作用及相互影響。數十年來,科學家們開發出多種策略,能夠在試管內甚至生物系統中對目標蛋白質進行修飾,以滿足不同的應用需求。然而,這些方法往往需要針對不同應用設計並合成專門的探針,即使是相同的目標蛋白質也不例外,這不僅耗時,還極大地消耗資源。
為解決這一問題,我們團隊開發了一種通用的兩階段(two-step)蛋白質功能化策略。在第一階段,團隊利用配體導向(ligand-directed)化學,在目標蛋白質上修飾可進行點擊化學的標記。隨後,在第二階段,透過生物正交點擊反應,將所需的功能基團與目標蛋白質結合,從而實現對其的探測與活性調控。
在這項研究中,團隊選擇碳酸酐酶(carbonic anhydrase)作為模型蛋白質進行功能化。他們 成功地利用螢光染料標記內源性碳酸酐酶,以進行細胞成像。此外,團隊還將一種光切換配 體與碳酸酐酶結合,使其活性能夠透過光響應方式進行調控,展現了該策略在動態調節蛋白 質功能方面的潛力;我們也正積極延伸此策略,使其能用於其他生理中重要蛋白質,並引入 光交聯/光解(photocrosslink/ photorelease)手段來加強第一階段標記的時空選擇性。
詳細研究成果請參閱:https://pubs.acs.org/doi/abs/10.1021/acschembio.3c00102
Deciphering the biological transport and functional interactions of proteins in their native environments has long been a major challenge in the field of biology. Chemical modifications of proteins provide valuable insights into their functions and interactions. Over the past decades, scientists have developed various strategies to modify target proteins in vitro and even in biological systems to meet diverse application needs. However, these approaches often require the design and synthesis of specific probes for different applications, even for the same target protein, making the process time-consuming and resource-intensive.
To address this issue, our team has developed a universal two-step protein functionalization strategy. In the first step, we utilize ligand-directed chemistry to introduce a clickable tag onto the target protein. In the second step, the desired functional moiety is conjugated to the target protein via bioorthogonal click reactions, enabling its detection and activity regulation.
In this study, we selected carbonic anhydrase as a model protein for functionalization. Our team successfully labeled endogenous carbonic anhydrase with a fluorescent dye for cellular imaging. Furthermore, we conjugated a photo-switchable ligand to carbonic anhydrase, allowing its activity to be regulated in a light-responsive manner. This demonstrates the potential of our strategy for dynamically modulating protein function. Additionally, we are actively extending this approach to other physiologically important proteins and incorporating photocrosslinking/photorelease techniques to enhance the spatiotemporal selectivity of the first-step labeling process.

For detailed research findings, please refer to: https://pubs.acs.org/doi/abs/10.1021/acschembio.3c00102