即時較高通量人類中樞神經膠質細胞髓鞘形成與退化系統之建立及於新穎疾病分子生物標記功能性分析之應用
Establishing Quantitative Higher Throughput Human Oligodendrocyte In Vitro (de)Myelination Platform for Functional Characterization of Multiple System Atrophy (MSA) Patients Derived Biomarkers
計畫主持人:臺大-生物科技所 林劭品、中研院-分生所 鄭珮琳
Prof. Shau-Ping Lin’s lab and collaborators initiated various oligodendrocyte based cell models for different levels of functional assays to evaluate the (Patent Pending, US 63/646,061) to characterize plasma microRNA, protein and extracellular vesicle protein biomarkers, which have been identified and validated in the collaborative team between National Taiwan University, National Taiwan University Hospital and Academia Sinica from various Parkinsonism patient groups (US-2024- 0327915-A1; US-2024-0331862-A1; ROC 113112010, Patents Pending, Technical reports published on 3rd of October, 2024).
The Cheng lab investigates Ecm29's role in neuroglial cells and its impact on EAE. Transcriptomic analysis of spinal cord tissue from EAE-affected mice with oligodendrocyte and microglial Ecm29 conditional knockout (cKO) revealed distinct gene expression changes. Prior to EAE induction, cKO oligodendrocytes and Schwann cells showed upregulated genes related to cell proliferation and differentiation, but not inflammation. At peak EAE, these cKO mice exhibited increased chemotaxisrelated gene expression and decreased neuronal activity gene expression, suggesting a link between Ecm29 loss and immune infiltration. Neuronal Ecm29 cKO mice showed minimal immune-related gene expression changes, consistent with their comparable EAE scores.
To explore the functional consequences of Ecm29 loss, an in vitro myelination model (Incorporated into the US 63/646,061 Pending Patent illustrated in the Figure attached) was used. Oligodendrocyte precursor cells were cultured on a neuronal substrate, inducing differentiation and myelin formation. MOG35-55-reactive lymphocytes were introduced, and their motility was tracked. Lymphocytes interacting with Ecm29 knockdown (KD) myelin, especially with IFN-γ treatment, showed increased trapping and slower movement compared to control myelin. Furthermore, Ecm29 KD myelinating oligodendrocytes exhibited increased binding to proliferating lymphocytes and enhanced immunological synapse formation. These in vitro findings suggest that Ecm29 KD myelin alters immune cell movement, potentially contributing to observed EAE progression changes.
國立臺灣大學林劭品老師實驗室與臺大醫院、中央研究院、成功大學合作夥伴建立了各種基 於寡突膠細胞的細胞模型,以進行不同層次的神經髓鞘功能性測試(如下圖所示; 專利審查 中,US 63/ 646,061),用於驗證血漿中的微型 RNA、蛋白質和細胞外囊泡蛋白生物標誌物之 生理或病理功能。此等生物標誌為先前本團隊與日本頂尖學者合作,於臺大醫院不同帕金森 氏症候群患者群體中解析出具有潛力之候選分子,並於此計畫執行期間於新收案之病人檢體 中驗證(專利審查中:US-2024-0327915-A1;ROC 113112010,US-2024-0331862-A1,技術報 告已經美國專利商標局於2024 年 10 月3 日公開在案)。
鄭珮琳老師實驗室(中研院)研究Ecm29 在神經膠質細胞中的作用及其對實驗性自身免疫性 腦脊髓炎(EAE)的影響。對 EAE 患病小鼠脊髓組織進行轉錄體分析,發現寡突膠質細胞和小 膠質細胞 Ecm29 條件性敲除(cKO)的小鼠表現出明顯的基因表達變化。在 EAE 誘導之前, cKO 寡突膠質細胞和許旺氏細胞顯示與細胞增殖和分化相關的基因上升,但與炎症無關。在 EAE 高峰期,這些 cKO 小鼠表現出趨化性相關基因表達增加和神經元活性基因表達減少,顯 示 Ecm29 缺失與免疫浸潤之間存在相關性。神經元 Ecm29 cKO 小鼠的免疫相關基因表達變 化極小,與其相似的EAE 評分一致。
為了探討 Ecm29 缺失的功能後果,我們使用體外髓鞘形成模型 (已納入審查中專利 US 63/646,061)。將寡突膠質細胞前體細胞培養在神經元基質上,誘導其分化和髓鞘形成。引入 MOG35-55 反應性淋巴細胞,並追蹤其運動。與對照髓鞘相比,與Ecm29 敲低(KD)髓鞘相互 作用的淋巴細胞,尤其是在IFN-γ處理下,表現出捕獲增加和運動減慢。此外,Ecm29 KD 髓 鞘化寡突膠質細胞表現出與增殖淋巴細胞的結合增加和免疫突觸形成增強。這些體外研究結 果表明,Ecm29 KD 髓鞘改變了免疫細胞的運動,可能導致觀察到的EAE 進程變化。
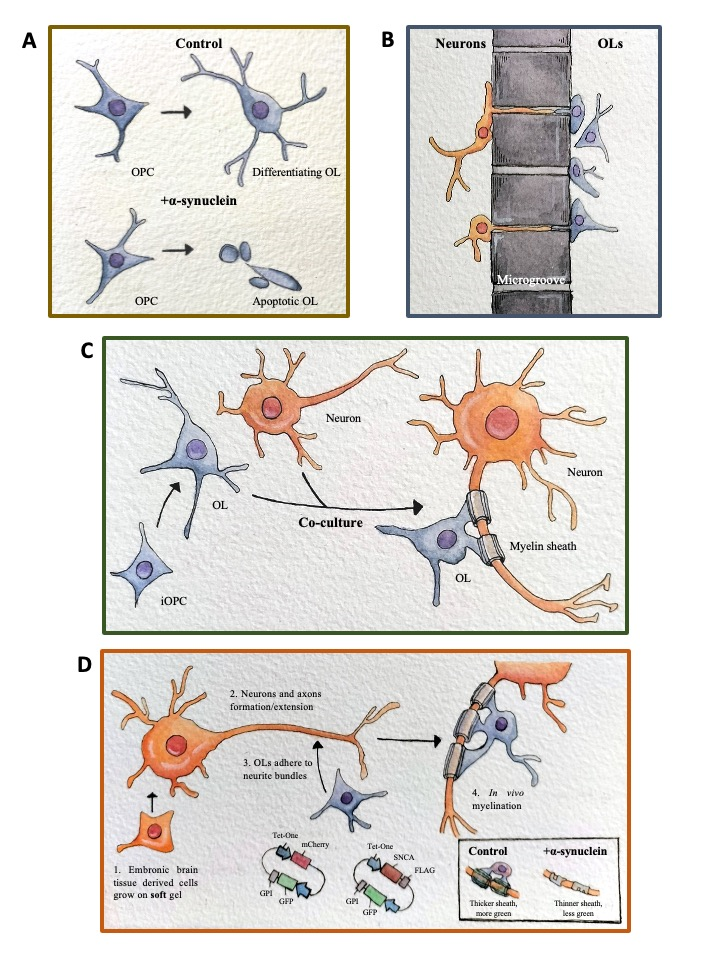
Figure. Establishment of multi-level in vitro myelination platform for functional assay and drug screening towards the understanding and mitigation of myelination defects. Various levels of in vitro quantitative/qualitative models were built to represent the function and properties of oligodendrocytes and their engagement of neuronal cells. (A) Several single cloned inducible alpha-Synuclein production Oligodendrocyte progenitor cell lines (MO3.13 based) and their microRNA/transcriptome profiling results are available at the undifferentiated and differentiating stages with or without alpha-Synuclein overexpression. Specific alpha-Synuclein responsive microRNA and transcripts are identified as possible drug screen readouts; (B) Microgroove containing multichannel co-culture between differentiating oligodendrocyte (MO3.13) and Neuronal cell line (SHSY5Y) are optimized for observation and quantify oligodendrocyte-neuronal interaction; (C) Co-culture of further differentiated oligodendrocytes (MO3.13) and neuronal cells (SHSY5Y) are optimized in 2D condition for observation of initial myelination event; (D) Real-time quantitative in vitro (de)myelination platform between rat neuronal lineage cells and rat or human transgenic oligodendrocyte progenitor cells. Functional analysis tested/being testing include the addition of conditioned medium or purified, labeled extracellular vesicles from alpha-Synuclein overexpressing cells and quantify the reduction of the thickness and area of myelinated neurites via the intensity of fluorescent signals specifically expressed in the cell membrane of the oligodendrocytes. This quantitative in vitro myelination platform has also been successfully used to reveal the mechanism underlying the prevention of autoimmune response in central nerve system (prevention of over proliferation of self-antigen recognizing effector T cells by Ecm29/Proteasome; Published in Cell Reports. 44: 115161, 2025).