揭示單細胞訊息系統的非線性特質
Reveal nonlinear properties of signaling systems in single cells
計畫主持人:臺大-海洋所 謝志豪、中研院-分生所 陳昇宏
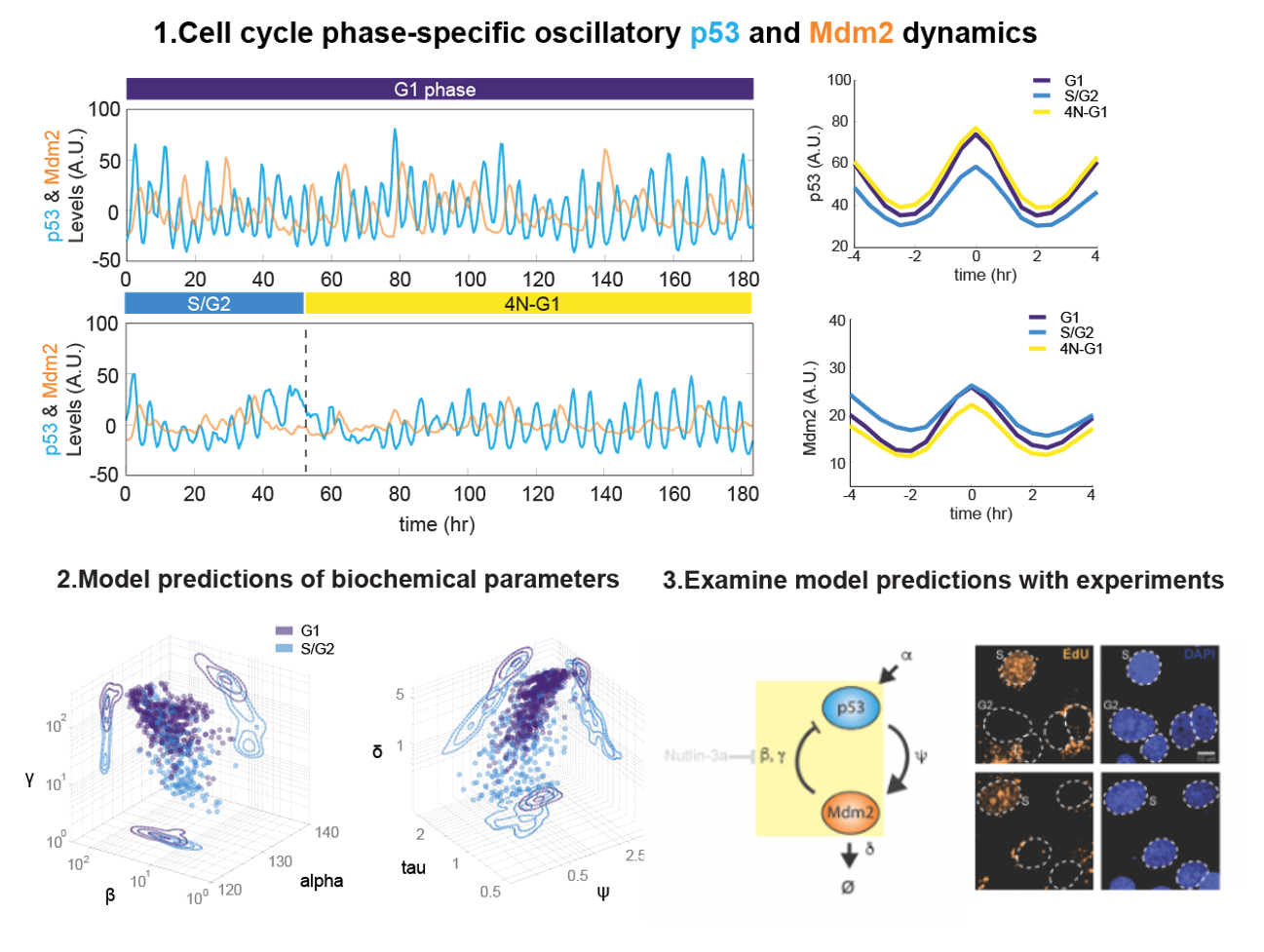
本計畫透過結合單細胞實驗技術與數學建模,深入探討單細胞內訊息傳遞系統的非線性特 性,著重於p53/Mdm2 調控機制在不同細胞週期階段的變化。研究結果顯示,在G1期細胞 中,p53 與Mdm2的降解速率顯著高於S/G2期,且p53 與Mdm2之間的親和力較弱,這些 細胞週期特定的參數對細胞內訊息振盪的形成具有關鍵作用。透過Nutlin-3a藥物干預及轉 錄抑制實驗,本研究利用單細胞追蹤技術精確量化了p53 及Mdm2的動態變化,並驗證了 模型預測。數據分析結果進一步揭示,這些參數變化可能影響細胞對DNA 損傷的反應,進 而調控細胞的生存與凋亡決策。研究成果不僅深化了我們對細胞內信號傳遞機制的理解, 也為未來針對p53 路徑失調所引發的疾病(如癌症)的治療提供了新思路。此外,本計畫 的跨院校合作模式促進了理論與實驗相結合的研究方法,為細胞動態調控機制的深入探索 奠定了堅實的理論與實踐基礎。總結來說,本研究成功驗證了細胞週期在訊息傳遞調控中 的關鍵角色,並展示了單細胞層級上非線性動態調控的潛在應用價值。
This project combines single-cell experimental techniques with mathematical modeling to deeply explore the nonlinear properties of intracellular signaling systems, with a particular focus on the variations in the p53/Mdm2 regulatory mechanism during different phases of the cell cycle. The research results indicate that in G1-phase cells, the degradation rates of both p53 and Mdm2 are significantly higher than in the S/G2 phase, and the binding affinity between p53 and Mdm2 is weaker. These cell cycle-specific parameters play a crucial role in the generation of intracellular signaling oscillations. By employing Nutlin-3a drug intervention and transcription inhibition experiments, the study precisely quantified the dynamic changes of p53 and Mdm2 using single-cell tracking techniques and validated the model predictions. Further data analysis revealed that these parameter variations might affect the cellular response to DNA damage, thereby regulating decisions related to cell survival and apoptosis. The research outcomes not only deepen our understanding of intracellular signal transduction mechanisms but also offer novel insights for the treatment of diseases (such as cancer) that arise from p53 pathway dysregulation. Moreover, the crossinstitutional collaborative model adopted in this project has fostered an integrated approach combining theoretical and experimental methods, thereby laying a solid foundation for further exploration of dynamic cellular regulation mechanisms. In summary, this study has successfully confirmed the critical role of the cell cycle in the regulation of signal transduction and demonstrated the potential applicability of nonlinear dynamic regulation at the single-cell level.